Accelerating the Path to Alzheimer's Prevention
Accelerating the Path to Alzheimer's Prevention
Accelerating the Science We Need for the Prevention We Want.
Lisa is an Alzheimer’s warrior, currently taking a drug developed through an ATRI collaboration to slow the progression of her disease.
The USC Epstein Family Alzheimer’s Research Institute leads global trials focused on treatments, prevention and ultimately a cure.

Paul Aisen, MD
Learn About the Rigorous Science Behind Our Trials and Why It Differentiates Us.
For more than a decade, ATRI has pioneered the most rigorous approach to Alzheimer’s clinical trials. Under Dr. Aisen’s leadership, the institute has built an unmatched infrastructure for designing and executing large-scale multicenter trials that are advancing the field toward prevention.
Dr. Aisen’s scientific philosophy centers on precision, collaboration, and the belief that Alzheimer’s disease can be prevented through early intervention and innovative trial design.
OUR MISSION AND VISION
TO PREVENT AND EFFECTIVELY TREAT ALZHEIMER’S DISEASE
1906
Today
1900-1970
First Discovery
1906
Dr. Alois Alzheimer first diagnosis
Alzheimer’s Disease got the name from Dr. Alois Alzheimer. He first noticed changes in the brain tissue of a woman who had died of an unusual mental illness. Her symptoms included memory loss, language problems, and unpredictable behavior. After she died, he examined her brain and found many abnormal clumps (now called amyloid plaques) and tangled bundles of fibers (now called neurofibrillary, or tau, tangles).
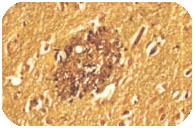
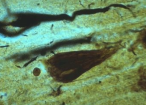
Alzheimer’s Disease got the name from Dr. Alois Alzheimer. He first noticed changes in the brain tissue of a woman who had died of an unusual mental illness. Her symptoms included memory loss, language problems, and unpredictable behavior. After she died, he examined her brain and found many abnormal clumps (now called amyloid plaques) and tangled bundles of fibers (now called neurofibrillary, or tau, tangles).
1906-1970
AD Assumed as Untreatable Degenerative Disease
1974-1986
Emerging Research
1974
Founding of National Institute on Aging (NIA)
1976
“The Prevalance and Malignancy of Alzheimer’s Disease” In Journal Archives of Neurology
Late 1970's
Cholinergic Hypothesis Suggests Treatment Possibilities
1984
Beta-Amyloid Identified as Prime Suspect in Triggering Nerve Cell Damage
1986
Tau Protein Identified a Key component of Tangles
1987-1994
Steps Toward Treatment
1987
First Encouraging Alzheimer’s Drug Trial
1991
Alzheimer’s Disease Cooperative Study (ADCS) Launched
1993
First Drug Approved
1994
Former U.S. President Ronald Reagan Alzheimer’s Diagnosis is Announced
We're the USC Epstein Family Alzheimer's Therapeutic Research Institute
For the past decade we’ve been on a path to solve the problem of Alzheimer’s Disease (AD). We have a roadmap for our research; built on amyloid treatments, add tau treatments and an array of persistent clinical studies all with one goal in mind — prevention.

The Alzheimer's Clinical Trials Consortium
Our Research + Laboratory Leaders
Paul Aisen,
Alison Belsha
Gustavo Jimenez-Maggiora, PhD, MBA
Gustavo Jimenez-Maggiora, PhD, MBA
Cecily Jenkins, PhD
Cecily Jenkins, PhD
Jeremy Pizzola
Michael Rafii, MD, PhD
Rema Raman, PhD
Rema Raman, PhD
Robert Rissman, PhD
Robert Rissman, PhD
Education
Impact AD
Educational initiative providing healthcare professionals with the latest evidence-based information on Alzheimer’s disease diagnosis and management.
Continuing Education
Professional development resources and training programs for researchers and clinicians in the field of Alzheimer’s and dementia.
